Publications:
135. Huang, J.; Tian, W.; Wang, S.; Yu, Y.; Yang, H.;* Tang, W;* "Palladium-Catalyzed Asymmetric Markovnikov Hydrocarbonylation of Vinyl Arenes".
CCS Chem. 2026, Jsut Accepted.
[doi]
134. Wang, N.; Yang, H.;* Tang, W;* "Enantioselective Ruthenium-Catalyzed Hydrogenation of Substituted 3-Acylamino Pyridinium Salts".
ACS Catal. 2026, 16, 7717-7725.
[doi]
133. Jin, J.; Li, X.; Luo, Y.; Chen, J;* Tang, W;* Du, K.* "Acyl-oxyallenes as α,β-unsaturated ketone surrogates for Giese radical addition".
Chem. Sci. 2026, 17, 2193.
[doi]
132. Yu, Y.; Zhang, Y.; Xue, X.-S.;* Hu, X.;* Tang, W.* "Enantiodivergent Rh-Catalyzed Reductive Hydroformylation of Alkenyl Boronic Esters".
Angew. Chem. Int. Ed. 2025, 65, e20602.
[doi]
131. Wang, N.; Tang, W.* "A sterically hindered phosphorus ligand for sterically hindered aryl-alkyl coupling reaction".
Chin. J. Org. Chem. 2025, 9, 3351-3360.
[doi]
130. Zhang, D.; Yang, H.;* Zhou Q.;* Tang, W.* "Enantioselective synthesis of chiral 2,3-cis-disubstituted piperidines and C1-substituted tetrahydroisoquinolines by asymmetric Cu-catalyzed cyclizative aminoboration".
Nat. Commun. 2025, 16, 6851.
[doi]
129. Chen, T.; Wang, H.-Y.; Xu, R.; Xu, G.; Yang, H.;* Sun, J.;* Chung, L. W.;* Tang, W.* "Asymmetric intramolecular reductive coupling of bisimines templated by chiral diborons".
Chem. Sci., 2025, 16, 13298-13305.
[doi]
128. Zhang, Z.; Feng, Y.; Chen, J.; Liu, X.;* Smith, M. D.;* Du, K.;* Tang, W.* "Visible Light-Promoted Construction of Cage-Like Frameworks by Sequential [2 + 2] Cycloaddition".
Org. Lett. 2025, 27, 7120-7125.
[doi]
127. Yu, Y.; Tang, Y.; Xu, S.; Liu, X.; Lv, Y.; Hu, X.;* Tang, W.* "Development of P‑Chiral Bisoxaphospholane Ligand BIOP and Its
Application in Rhodium-Catalyzed Asymmetric Hydroformylation".
Org. Lett. 2025, 27, 6251-6256.
[doi]
126. Wang, Z.; Guo, S.; Shen, Z.; Wei, R.; Jiang, R.; Liu, G.; Zhou, Q.;* Du, K.;* Tang, W.* "Enantioselective palladium-catalyzed α-arylation of primary alkylamines".
Chem Catalysis 2025, 5, 101331.
[doi]
125. Huang, J. (1st co-author); Pei, C. (1st co-author); Yang, H.;* Wu, B.;* Tang, W.* "Enantioselective Palladium-Catalyzed α-Arylation of Acyclic Esters".
Angew. Chem. Int. Ed. 2025, 64, e202505458.
[doi]
124. Cao, Z.*; He, D.; Luo, L.; Tang, W.* "Recent Advances in Enantioselective Transition Metal Catalysis Mediated by Ligand–Substrate Noncovalent Interactions".
Catalysts 2025, 15, 395.
[doi]
123. Fu, X.; Yu, H.; Gao, H.; Dai, Y.; Yang, H.; Tang, W.* "A chiral ferrocene-tethered ruthenium diamine catalyst for asymmetric transfer hydrogenation of ketones".
Org. Chem. Front., 2025, 12, 2957-2962.
[doi]
122. Zhu, Y. (1st co-author); Zhang, Y. (1st co-author); He, D.; Yang, H.;* Xue, X.-S.;* Tang, W.* "Rhodium-Catalyzed Asymmetric Reductive Hydroformylation of α-Substituted Enamides".
J. Am. Chem. Soc. 2024, 146, 33249-33257.
[doi]
121. Luo, B.; Dong, W.; Ma, Q.; Yang, H.;* Tang, W.* "Synthesis of Biheteroaryls by Pd-Catalyzed Homocoupling of Heteroaryl Bromides".
Org. Lett. 2024, 26, 8736-8740.
[doi]
120. Lin, Y.; Xu, G.;* Tang, W.* "Chiral Polymeric Diamine Ligands for Iridium-Catalyzed Asymmetric Transfer Hydrogenation".
J. Am. Chem. Soc. 2024, 146, 27736-27744.
[doi]
119. Lv, Y.; Liao, H.; Tang, W.* "Synthesis of a Sitagliptin Intermediate via Ruthenium-Catalyzed Asymmetric Hydrogenation of Enamine Ester".
Chin. J. Org. Chem. 2024, 44, 3720-3726.
[doi]
118. Dong, W.; Wang, N.; Yang, H.; Xu, G.; Tang, W.* "A Polymeric Phosphorus Ligand for the Synthesis of Herbicide Rinskor".
Acta Chimica Sinica 2024, 82, 940-953.
[doi]
117. Cao, Z.; He, D.; Tang, W.* “Catalysis and Synthesis Enabled by P-Chiral Dihydrobenzooxaphosphole Ligands”.
Org. Process Res. Dev. 2024, 28, 949-977. [doi] |
116. Bai, H. (1st co-author); Gu, W.(1st co-author); Zhao, D.; Xu, G.;* Tang, W.* “Synthesis of dydrogesterone by aromatization-dearomatization strategy”.
Green Synthesis and Catalysis. 2023, 6, 267-274. [doi] |
115. Peng, H.(1st co-author); Zhao, D.(1st co-author); Tang, W.;* Peng, A.* “Dienediamine: A Safe Herbicide as Paraquat Surrogate”.
Mol. Plant. 2023, 16, 1962-1975. [doi] |
114. Liu, Z.; Gao, B.; Chernichenko, K.; Yang, H.;* Lemaire, S.;* Tang, W.* “Enantioselective C-H Arylation for Axially Chiral Heterobiaryls”.
Org. Lett. 2023, 25, 7004-7008. [doi] |
113. Yang, H.(1st co-author); Yu, H.(1st co-author); Stolarzewicz. I. A.;* Tang, W.* “Enantioselective transformations in the synthesis of therapeutic agents”.
Chem. Rev. 2023, 123, 9397-9446. [doi] |
112. Sun, J.; Chen, Y.; Ragab. S. S.; Gu, W.; Tang, Z.; Tang, Y.; Tang, W.* “Total Syntheses of Polyhydroxylated Steroids by an Unsaturation-Functionalization Strategy”.
Angew. Chem., Int. Ed. 2023, 62, e202303639. [doi] |
111. Zhou, M.(1st co-author); Lin, Y.(1st co-author); Chen, X.-X.(1st co-author); Xu, G.;* Chung, L. W.;* Tang, W.* "Asymmetric Synthesis of Vicinal Tetrasubstituted Diamines via Reductive Coupling of Ketimines Templated by Chiral Diborons".
Angew. Chem., Int. Ed. 2023, 62, e202300334. (highlighted as Angew. Chem., Int. Ed. Hot Paper) [doi] |
110. Zhu, Y.; Dong, W.; Tang, W.* "Palladium-Catalyzed Cross-Couplings in the Synthesis of Agrochemicals".
Advanced Agrochem 2022, 1, 125-138. [doi] |
109. Li, B.; Luo, B.; Yang, H.;* Tang, W.* "Heck Reaction of N-Heteroaryl Halides for Concise Synthesis of Chiral α-Heteroaryl-substituted Heterocycles".
Angew. Chem., Int. Ed. 2022, 61, e202209087. [doi] |
108. Yang, H.; Tang, W.* “Enantioselective Construction of ortho-Sulfur- or Nitrogen-Substituted Axially Chiral Biaryls and Asymmetric Synthesis of Isoplagiochin D”.
Nat. Commun. 2022, 13, 4577. [doi] |
107. Kang, X.; Qian, C.; Yang, H.; Shi, J.;* Claverie, J.;* Tang, W.* “Protecting-group-free enantioselective tandem allylic substitution of o-phenylenediamines and o-aminophenols”.
Green Synthesis and Catalysis. 2022, 3, 185-189. [doi] |
106. Li, Z.; Xu, R.; Guo, H.; Yang, H.; Xu, G.; Shi, E.* Xiao, J.* Tang, W.* “Enantioselective Rhodium-Catalyzed Hydrogenation of (Z)‑N‑Sulfonyl-α-Dehydroamido Boronic Esters”.
Org. Lett. 2022, 24, 714-719. [doi] |
105. Li, B.; Luo, B.; Blakemore, C. A.;* Smith, A. C.; Widlicka, D. W.; Berritt, S.; Tang, W.* “Synthesis of α-Heteroaryl Propionic Esters by Palladium-Catalyzed α-Heteroarylation of Silyl Ketene Acetals”.
Org. Lett. 2021, 23, 6439-6443. [doi] |
104. Tian, W.; Li, B.; Tian, D.; Tang, W.* “Regioselective 2-alkylation of indoles with α-bromo esters catalyzed by Pd/P,P=O system”.
Chin. Chem. Lett. 2022, 33, 197-200. [doi] |
103. Sun, J.; Gu, W.; Yang, H.;* Tang, W.* “Enantioselective total synthesis of parnafungin A1 and 10a-epi-hirtusneanine”.
Chem. Sci. 2021, 12, 10313-10320. [doi] |
102. Wan, F.; Wang, N.; Zhu, Y.; Tang, C.; Claverie, J. P.;* Tang, W.* “Enantioselective Hydrogenation of Cyclic Tetrasubstituted-Olefinic Dehydroamino Acid Derivatives”.
Chem. Commun. 2021, 57, 5546-5549. [doi] |
101. Peng, H.; Li, T.; Tian, D.; Yang, H.; Xu, G.; Tang, W.* “Metal-Free Reduction of Unsaturated Carbonyls, Quinones, and Pyridinium Salts with Tetrahydroxydiboron/Water”.
Org. Biomol. Chem. 2021, 19, 4327-4337. [doi] |
100. Xu, R.; Gao, Z.,; Yu, Y.; Tang, Y.; Tian, D.; Chen, T.; Chen, Y. ; Xu G. Shi, E.; Tang, W.* "A Facile and Practical Preparation of P-Chiral Phosphine Oxides".
Chem. Commun. 2021, 57, 3335-3338. [doi] |
99. Wu, T.; Zhou, Q.; Tang, W.* “Enantioselective α‐Carbonylative Arylation for Facile Construction of Chiral Spirocyclic β,β'‐Diketones".
Angew. Chem., Int. Ed. 2021, 60, 9978-9983. [doi] |
98. Sun, J.; Yang, H.;* Tang, W.* "Recent advances in total syntheses of complex dimeric natural products".
Chem. Soc. Rev. 2021, 50, 2320-2336. [doi] |
97. Tian, D,; Xu, R,; Zhu, J.; Huang, J.; Dong, W.; Claverie, J.; Tang, W.* "Asymmetric Hydroesterification of Diarylmethyl Carbinols".
Angew. Chem., Int. Ed. 2021, 60, 6305-6309. [doi] |
96. Tang, W.* "Author Profile".
Angew. Chem., Int. Ed. 2020, 59, 7988. [doi] |
95. Wan, F.; Tang, W.* "Phosphorus Ligands from the Zhang Lab: Design, Asymmetric Hydrogenation, and Industrial Applications".
Chin. J. Chem. 2021, 39, 954-968. [doi] |
94. Aliyu, M.; Li, B.; Yang, H.; Tang, W.* "Palladium-catalyzed reductive cross-coupling between α-bromo carboxamides and terminal alkynes".
Org. Chem. Front. 2020, 7, 3505-3508. [doi] |
93.Li, K.; Nie, M.; Tang, W.* "Synthesis of α-tertiary allylsilanes by palladium-catalyzed hydrosilylation of 1,1-disubstituted allenes".
Green Synthesis and Catalysis. 2020, 1, 171-174. [doi] |
92 Yu, H.; Yang, H.;* Shi, E.;* Tang, W.* “Development and Clinical Application of Phosphorus-Containing Drugs".
Med. Drug Discov. 2020, 8, 100063. [doi] |
91.Wu, T.; Tang, W.* “Construction of Bridged Polycyclic Skeletons via Transition‐Metal Catalyzed Carbon‐Carbon Bond‐Forming Reaction".
Chem. Eur. J. 2021, 27, 3944-3956. [doi] |
90.Li, B.; Aliyu, M.; Gao, Z.; Li, T.; Dong, W.; Shi, E.* Tang, W.* “General Synthesis of Chiral α,α-Diaryl Carboxamides by Enantioselective Palladium-Catalyzed Cross-Coupling".
Org. Lett. 2020, 13, 4974-4978. [doi] |
89.Wu, T.; Kang, X.; Bai, H.; Xiong, W.; Xu, G.; Tang, W.* “Enantioselective Construction of Spiro Quaternary Carbon Stereocenters via Pd-Catalyzed Intramolecular α-Arylation".
Org. Lett. 2020, 22, 4602-4607. [doi] |
88. Qian, C.; Tang, W.* "A Versatile Synthesis of Vinyl-Substituted Heterocycles via Regio- and Enantioselective Pd-Catalyzed Tandem Allylic Substitution".
Org. Lett. 2020, 22, 4483-4488. [doi] |
87. Zhou, M.; Li, K.; Chen, D.; Xu, R.; Xu, G.* Tang, W.* "Enantioselective Reductive Coupling of Imines Templated by Chiral Diboron".
J. Am. Chem. Soc. 2020, 142, 10337-10342. [doi] |
86. Xu, R.H.; Yang, H.; Tang, W.*“Efficient Synthesis of Chiral Drugs Facilitated by P-Chiral Phosphorus Ligands”.
Chin. J. Org. Chem. 2020, 14, 581. [doi] |
85. Liu, Z.; Li, T.; Zhao, J.; Tang, W.* “A substituted tricyclohexylphosphane with ‘conformational lock’”.
Tetrahedron. 2020, 76, 131216. [doi] |
84. Yang, H.; Sun, J.; Gu, W.; Tang, W.* "Enantioselective Cross-Coupling for Axially Chiral Tetra-ortho-Substituted Biaryls and Asymmetric Synthesis of Gossypol".
J. Am. Chem. Soc. 2020, 142, 8036–8043. [doi] |
83. Mu, X.; Yu, H.; Peng, H.; Xiong, W.; Wu, T.; Tang, W.* "Expedite Construction of Various Bridged Polycyclic Skeletons by Palladium-Catalyzed Dearomatization".
Angew. Chem., Int. Ed. 2020, 59, 8143-8147. [doi] |
82. Wu, T; Xu, G; Tang, W.* “P-Chiral Phosphorus Ligands for Cross-Coupling and Asymmetric Hydrogenation Reactions”.
Aldrichimica Acta 2020, 53, 27-35. [doi] |
81. Li, C.; Ragab. S.S.; Liu, G.;* Tang, W.* "Enantioselective Formation of Quaternary Carbon Stereocenters in Natural Product Synthesis: A Recent Update".
Nat. Prod. Rep. 2020, 37, 276. [doi] |
80. Zhu, J.; Huang, L.; Dong, W.; Li, N.; Yu, X.; Deng, W.*; Tang,W.* "Enantioselective Rhodium-Catalyzed Addition of Arylboroxines to N-Unprotected Ketimines: Efficient Synthesis of
Cipargamin".
Angew. Chem., Int. Ed. 2019, 58, 16119-16123. (highlighted as Angew. Chem., Int. Ed. VIP Paper) [doi] |
79. Li, C.(1st co-author); Wan, F.(1st co-author); Chen, Y.; Peng, H.; Tang, W.*; Yu, S.*; McWilliams, J. C.*, Mustakis, J.; Samp, L.; Maguire, R. J. "Stereoelectronic Effects in Ligand Design: Enantioselective RhodiumCatalyzed Hydrogenation of Aliphatic Cyclic Tetrasubstituted Enamides and Concise Synthesis of (R)-Tofacitinib".
Angew. Chem., Int. Ed. 2019, 58, 13573-13583. [doi] |
78. Li, B.; Li, T.; Aliyu, M. A.; Li, Z.; Tang, W.* "Enantioselective Palladium-Catalyzed Cross-Coupling of a-Bromo Carboxamides and Aryl Boronic Acids".
Angew. Chem., Int. Ed. 2019, 58, 11355-11359. [doi] |
77. Xiong, W.; Xu, G.; Yu, X.;* Tang, W.* "P‑Chiral Monophosphorus Ligands for Asymmetric CopperCatalyzed
Allylic Alkylation".
Organometallics 2019 , Just Accepted [doi] |
76. Xu, G.; Senanayake, C. H.; Tang, W.* "P‑Chiral Phosphorus Ligands Based on a 2,3-Dihydrobenzo[d][1,3]oxaphosphole Motif for Asymmetric Catalysis".
Acc. Chem. Res. 2019, 52, 1101−1112. [doi] |
75. Yang, H; Tang, W.* "Efficient Enantioselective Syntheses of Chiral Natural Products Facilitated by Ligand Design".
Chem. Rec. 2019, 19, 1-19. [doi] |
74. Dong, W; Xu, G; Tang, W.* "Enantioselective palladium-catalyzed C(sp2)-H carbamoylation".
Tetrahedron 2019, 75, 3239-3247. [doi] |
73. Liu, G.;Fu, W.;Mu, X.;Wu, T.;Nie, M.;Li, K.;Xu, X.; Tang, W.* "Pyrrolidines and Piperidines Bearing Chiral Tertiary Alcohols by Nickel-Catalyzed Enantioselective Reductive Cyclization of N-alkynones".
Commun. Chem. 2018, 1, 90-96. [doi] |
72. Wu, Z.;Si, T.;Xu, G.;Xu, B.;Tang, W.* "Ligand-Free Nickel-Catalyzed Kumada Couplings of Aryl Bromides with tert-Butyl Grignard Reagents".
Chinese Chemical Letters 2018, 30, 597–600. [doi] |
71. Tian, D.; Peng, H.; Liu, Z.; Tang, W.* "Optically Active N-Alkyl Aziridines via Stereospecific Reductive Cyclization of α-Mesylated Acetamides".
Org. Chem. Front. 2018, 5, 2723–2727. [doi] |
70. Rao, X(1st co-author).; Li, N(1st co-author).; Bai, H(1st co-author).; Wang, Z.; Tang, W.* "Efficient Synthesis of (-)-Corynoline by Sterically Hindered Enantioselective Palladium-Catalyzed α-Arylation".
Angew. Chem., Int. Ed. 2018, 55, 5044–5048. [doi] |
69. Tian, D(1st co-author).; Li, C(1st co-author).; Gu, G.; Peng, P.; Zhang, X.;* Tang, W.* "Stereospecific Nucleophilic Substitution with Arylboronic Acids as Nucleophiles: A Crucial CONH Effect".
Angew. Chem., Int. Ed. 2018, 55, 5044–5048. [doi] |
68. Jiang, W.; Zhao, Q.; Tang, W.* Efficient P‐Chiral Biaryl Bisphosphorus Ligands for Palladium‐Catalyzed Asymmetric Hydrogenation".
Chin. J. Chem. 2018, 36, 153-156. [doi] |
67. Si, T.; Li, B.; Xiong, W.; Xu, B.;* Tang, W.* "Efficient cross-coupling of aryl/alkenyl triflates with acyclic secondary alkylboronic acids".
Org. Biomol. Chem. 2017, 15, 9903-9909. [doi] |
66. Du, K(1st co-author).; Yang, H(1st co-author).; Guo, P(1st co-author).; Feng, L.;Xu, G.; Zhou, Q.; Chung, L, -W.; Tang, W.* "Efficient syntheses of (−)-crinine and (−)-aspidospermidine, and the formal synthesis of (−)-minfiensine by enantioselective intramolecular dearomative cyclization".
Chem. Sci. 2017, 8, 6247-6256. [doi] |
65. Chen, D.; Xu, G.; Zhou, Q.; Chung, L, -W.; Tang, W.* "Practical and Asymmetric Reductive Coupling of Isoquinolines Templated by Chiral Diborons".
J. Am. Chem. Soc. 2017, 139, 9767-9770. [doi] |
64. Liu J(1st co-author).; Nie, M(1st co-author).; Zhou, Q.; Gao, S.; Jiang, W.; Chung, L, -W*.; Tang, W*.; Ding, K.* "Enantioselective Palladium-Catalyzed Diboration of 1,1-Disubstituted Allenes".
Chem. Sci. 2017, 8, 5161-5165. [doi] |
63. Yang, H.; Yang, X.; Tang, W.* "Transition-metal catalyzed asymmetric carbon-carbon cross-coupling with chiral ligands".
Tetrahedron 2016, 72, 6143-6174. [doi] |
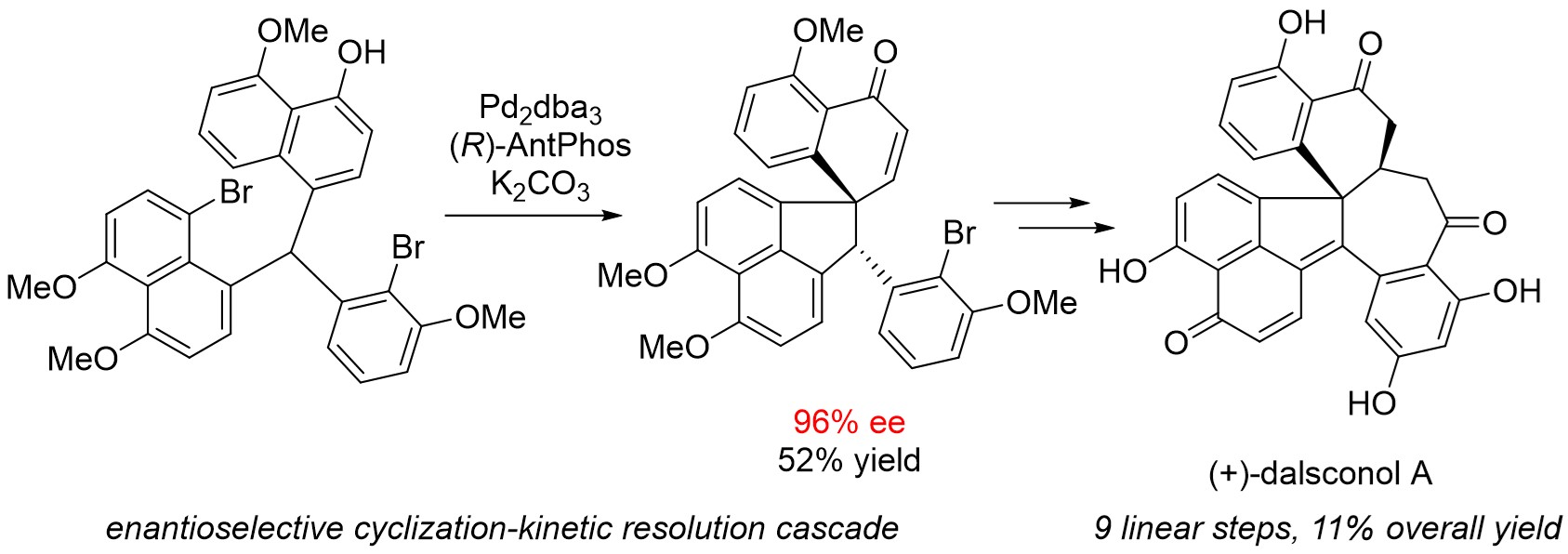
62. Zhao, G(1st co-author).; Xu, G(1st co-author).; Qian, C.; Tang, W.* "Efficient Enantioselective Syntheses of (+)-Dalesconol A and B".
J. Am. Chem. Soc. 2017, 139, 6630-6636. [doi] |
61. Li, C.; Zhang, Y.; Sun, Q.; Gu, T.; Peng, H.; Tang, W.* "Transition-Metal-Free Stereospecific Cross-Coupling with Alkenylboronic Acids as Nucleophiles".
J. Am. Chem. Soc. 2016, 138, 10774-10777. [doi] |
60. Li, C.; Chen, D.; Tang, W.* "Addressing the Challenges in Suzuki–Miyaura Cross-Couplings by Ligand Design".
Synlett. 2016. 27, 2183-2200. [doi] |
59. Fu, W.; Tang, W.* "Chiral Monophosphorus Ligands for Asymmetric Catalytic Reactions".
ACS. Catal. 2016, 6, 4814-4858. [doi] |
|
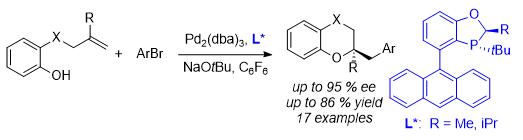
58. Hu, N.; Li, K.; Wang, Z.; Tang, W.* Synthesis of Chiral 1,4-Benzodioxanes and Chromans by Enantioselective Palladium-Catalyzed Alkene Aryloxyarylation Reactions".
Angew. Chem., Int. Ed. 2016, 55, 5044–5048. [doi] |
|
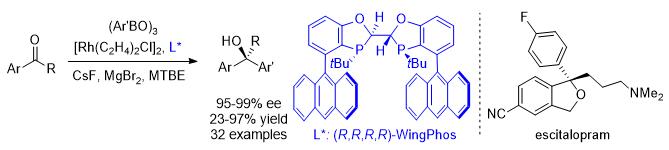
57. Huang, L.; Zhu, J.; Jiao, G.; Wang, Z.; Yu, X.; Deng, W.-P.;* Tang, W.* "Highly Enantioselective Rhodium-Catalyzed Addition of Arylboroxines to Simple Aryl Ketones: Efficient Synthesis of Escitalopram".
Angew. Chem., Int. Ed. 2016 55, 4527–4531
[doi]
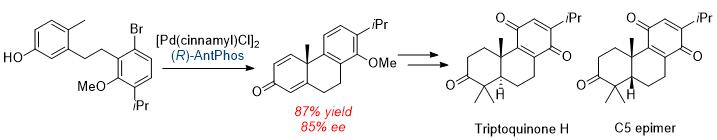
56. Cao, Z.; Du, K.; Liu, J.;* Tang, W.* "Synthesis of Triptoquinone H and its C-5 Epimer via Efficient Asymmetric Dearomative Cyclization".
Tetrahedron. 2016, 72, 1782–1786. (Invited Paper) [doi] |
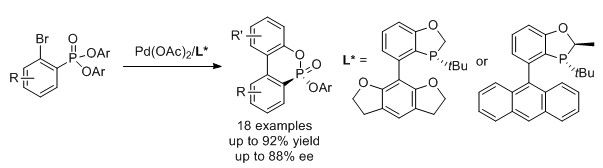
55. Yang, X.; Xu, G.; Tang, W.* "Efficient synthesis of chiral biaryls via asymmetric Suzuki-Miyaura cross-coupling of ortho-bromo aryl triflates".
Tetrahedron. 2016, 72, 5178–5183. (Invited paper). [doi]
|
|
|
|---|
54. Xu, G.; Li, M.; Wang, S.; Tang, W*. "Efficient synthesis of P-chiral biaryl phosphonates by stereoselective intramolecular cyclization".
Org. Chem. Front. 2015, 2, 1342-1345. [doi]
|
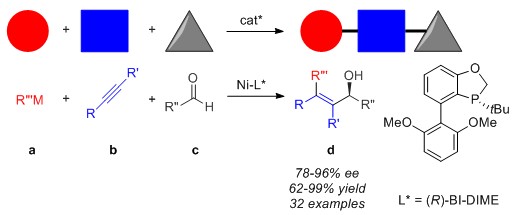
53. Nie, M.; Fu, W.; Cao, Z.; Tang, W*. "Enantioselective nickel-catalyzed alkylative alkyne–aldehyde cross-couplings".
Org. Chem. Front. 2015, 2, 1322-1325. [doi]
|
52. Hu, N(1st co-author).; Zhao, G(1st co-author).; Zhang, Y.; Liu, X.; Li, G.; Tang, W*. "Synthesis of Chiral α-Amino Tertiary Boronic Esters by Enantioselective hydroboration of α-Arylnamides".
J. Am. Chem. Soc. 2015, 137, 6746-6749. [doi]
|
| |
51. Du, K.; Guo, P.; Chen, Y.; Cao, Z.; Wang, Z,; Tang, W*. "Enantioselective Palladium-Catalyzed Dearomative Cyclization for Efficient Synthesis of Terpenes and Steroids".
Angew. Chem., Int. Ed. 2015, 54, 3033-3037. (hot paper). [doi]
50. Li, C.; Chen, T.; Li, B.; Xiao, G.; Tang, W*. "Efficient Syntheses of Sterically Hindered Acyclic Secondary Alkyl Arenes by Suzuki-Miyaura Cross-Couplings". Angew. Chem., Int. Ed. 2015, 54, 3792-3796. [doi]
49. Fu, W.; Nie, M.; Wang, A.; Tang, W*. "Highly Enantioselective Nickel-Catalyzed Intramolecular Reductive Cyclization of Alkynones". Angew. Chem., Int. Ed. 2015, 54, 2520-2524 . [doi]
48. Xu, G.; Zhao, Q.; Tang, W*. "Development of Efficient Asymmetric Suzuki-Miyaura Cross-Coupling and Applications in Synthesis".
Chin. J. Org. Chem. 2014, 34, 1919-1940 (Invited Account, Cover Paper). [doi]
47. Li, C.; Xiao, G.; Zhao, Q.; Liu, H.; Wang T.; Tang, W*. "Sterically Demanding Aryl-Alkyl Suzuki-Miyaura Coupling".
Org. Chem. Front. 2014, 1, 225-229 ( highlighted as OCF Cover Paper). [doi]
46. Xu, G.; Fu, W.; Liu, G.; Senanayake, C. H.; Tang, W*. "Efficient Syntheses of Korupensamines A, B and Michellamine B by Asymmetric Suzuki-Miyaura Coupling Reactions".
J. Am. Chem. Soc. 2014, 136, 570–573( highlighted as JACS Cover Paper, by Synfacts). [doi]
45. Reeves, J. T.*; Tan, Z.; Reeves, D. C.; Song, J. J.; Han, Z. S.; Xu, Y.; Tang, W.; Yang, B.; Razavi, H.; Harcken, C.; Kuzmich, D.; Mahaney, P. E.; Lee, H.; Busacca, C. A.; Senanayake, C. H. "Development of an Enantioselective Hydrogenation Route to (S)-1-(2-(Methylsulfonyl)pyridin-4-yl)propan-1-amine".
Org. Process Res. Dev. 2014, 18, 904–911. [doi]
44. Cai, Z.; Liu, G.; Jiao, G.; Senanayake, C. H.; Tang, W*. "Practical Syntheses of N-Acetyl (E)-β-Arylenamides".
Synthesis. 2013, 45, 3355-3360. [doi]
43. Liu, G.; Xu, G.; Luo, R.; Tang, W*. "Search for Ideal P-Chiral Phosphorus Ligands for Practical Asymmetric Hydrogenation and Asymmetric Suzuki-Miyaura Coupling".
Synlett. 2013, 24, 2465-2471 ( Synpacts, invited review). [doi]
42. Li, K.; Hu, N.; Luo, R.; Yuan, W.; Tang, W*. "A Chiral Ruthenium –Monophosphine Catalyst for Asymmetric Addition of Arylboronic Acids to Aryl Aldehydes". J. Org. Chem. 2013, 78, 6350-6355 ( highlighted by Synfacts). [doi]
41. Luo, R.; Li, K.; Hu, Y.; Tang, W*. "Enantioselective Rhodium-Catalyzed Addition of Arylboronic Acids to Trifluoromethyl Ketones". Adv. Syn. Cat. 2013, 355, 1297-1302. [doi]
40. Liu, G.; Liu, X.; Cai, Z.; Jiao, G.; Xu, G.; Tang, W*. "Design of Phosphorus Ligands with Deep Chiral Pockets: Practical Synthesis fo Chiral β-Arylamines by Asymmetric Hydrogenation".
Angew. Chem., Int. Ed. 2013, 52, 4235-4238 ( Highlighted in Chin. J. Org. Chem.; Chemistry Portal Highlights). [doi]
39. Zhao, Q.; Li, C.; Senanayake, C. H.; Tang, W*. "An Efficient Method for Sterically Demanding Suzuki-Miyaura Coupling Reactions". Chem. Eur. J. 2013, 19, 2261-2265. [doi]
38. Li, W.*; Rodriguez, S.; Duran, A.; Sun, X.; Tang, W.*; Premasiri, A.; Wang, J.; Sidhu, K.; Patel, N. D.; Savoie, J.; Qu, B.; Lee, H.; Haddad, N.; Lorenz, J.C.; Nummy, L.; Hossain, A.; Yee, N.; Lu, B.; Senanayake, C. H. "The P-Chiral Phosphane Ligand (MeO-BIBOP) for Efficient and Practical Large-Scale Rh-Catalyzed Asymmetric Hydrogenation of N-Acetyl Enamides with High TONs". Org. Process Res. Dev. 2013, 17, 1061–1065. [doi]
37. Tang, W.; Patel, N. D.; Wei, X.*; Byrne, D.; Chitroda, A.; Narayanan, B.; Sienkiewicz, A.; Nummy, L. J.; Sarvestani, M.; Ma, S.; Grinberg, N.; Lee, H.; Kim, S.; Li, Z.; Spinelli, E.; Yang, B.; Yee, N.; Senanayake, C.H. "Synthesis of a Sodium–Hydrogen Exchange Type 1 Inhibitor: An Efficient Cu-Catalyzed Conjugated Addition of a Grignard Reagent to an Acetyl Pyridinium Salt". Org. Process Res. Dev. 2013, 17, 382–389. [doi]
36. Tang, W.*; Patel, N. D.; Xu, G.; Xu, X.; Savoie, J.; Ma, S.; Hao, M.; Keshipeddy, S.; Capacci, A. G.; Wei, X.; Zhang, Y.; Gao, J. J.; Li, W.; Rodriguez, S.; Lu, B. Z.; Yee, N. K.; Senanayake, C.H. "Efficient Chiral Monophosphorus Ligands for Asymmetric Suzuki–Miyaura Coupling Reactions".
Org. Lett. 2012, 14 , 2258–2261. [doi]
35. Tang, W.*; Wei, X.; Yee, N. K.; Patel, N.; Lee, H.; Senanayake, C.H. "A Practical Asymmetric Synthesis of Isopropyl (1R,2S)-Dehydrocoronamate".
Org. Proc. Res. Dev. 2011, 15, 1207-1211. [doi]
34. Li, W.*; Gao, J. J.; Zhang, Y.; Tang, W.; Lee, H.; Fandrick, K. R.; Lu, B.; Senanayake, C.H. "A Mild Palladium-Catalyzed Suzuki Coupling Reaction of Quinoline Carboxylates with Boronic Acid".
Adv. Syn. Cat. 2011, 353, 1671-1675. [doi]
33. Tang, W.*; Patel, N. D.; Capacci, A. G.; Wei, X.; Yee, N. K.; Senanayake, C.H. "A Facile Synthesis of N-Acetyl Enamides by Reductive Acetylation of Oximes Mediated with Ferrous Acetate:Synthesis of N-(1-(4-Bromophenyl)vinyl)acetamide".
Org. Syn. 2013, 90, 62-73. [doi]
32.Tang, W.*; Keshipeddy, S.; Zhang, Y.; Wei, X.;Savoie, J.; Patel, N. D.; Yee, N. K.; Senanayake, C.H. "Efficient Monophosphorus Ligands for Palladium-Catalyzed Miyaura Borylation".
Org. Lett. 2011, 13, 1366-1369. [doi]
31. Rodriguez, S.*; Qu, B.; Haddad, N.; Reeves, D.; Tang, W.*; Krishnamurthy, D.; Senanayake, C.H. "Dihydrobenzooxaphosphole-Based Monophosphorus Ligands for Palladium-Catalyzed Amination Reactions".
Adv. Syn. Cat. 2011, 353, 533-537. [doi]
30. Tang, W.*; Wei, X.; Li, W.; White, A.; Patel, N. D.; Savoie, J.; Gao, J. J.' Rodriguez, S.; Qu, B.; Haddad, N.; Lu, B. Z.; Krishnamurthy, D.; Yee, N. K.; Senanayake, C.H. "A General and Special Catalyst for Suzuki-Miyaura Coupling Processes".
Angew. Chem., Int. Ed. 2010, 49, 5879-5883. ( Highlighted as "Synfact of the month" by Synfacts in Nov. 2010). [doi]
29. Fandrick, D. R.*; Fandrick, K. R.; Reeves, J. T.; Tan, Z.; Tang, W.; Capacci, A. G.; Rodriguez, S.; Song, J. J.; Lee, H.; Yee, N. K.; Senanayake, C.H. "Copper Catalyzed Asymmetric Propargylation of Aldehydes".
J. Am. Chem. Soc. 2010, 132, 7600-7601. [doi]
28. Tang, W.*; Capacci, A. G.; White, A.; Ma S.; Rodriguez, S.; Qu, B.; Savoie, J.; Patel, N.; Wei, X.; Haddad, N.; Grinberg, N.; Yee, N. K.; Krishnamurthy, D.; Senanayake, C. H. "Novel and Efficient Chiral Bisphosphorus Ligands for Rhodium-Catalyzed Asymmetric Hydrogenation".
Org. Lett. 2010, 12, 1104-1107. ( Highlighted by Synfacts). [doi]
27. Tang, W.*; Qu, B.; Capacci, A.; Rodriguez, S.; Wei, X.; Haddad, N.; Narayanan, B.; Ma S.; Grinberg, N.; Yee, N. K.; Senanayake, C. H. "Novel, Tunable, and Efficient Chiral Bisdihydrobenzooxaphosphole Ligands for Asymmetric Hydrogenation".
Org. Lett. 2010, 12, 176-179. [doi]
26. Ma S.; Shen, S.; Haddad, N.; Tang, W.; Wang, J.; Lee, H.; Yee, N.; Senanayake, C. H.; Grinberg, N.* "Chromatographic and Spectroscopic Studies on the Chiral Recognition of Sulfated beta-Cyclodextrin as Chiral Mobile Phase Additive".
J. Chromatography A, 2009, 1216, 1232-1240. [doi]
25. Tang, W.*; Capacci, A.; Sarvestani, M.; Wei, X.; Yee, N. K.; Senanayake, C. H. "A Facile and Practical Synthesis of N-Acetyl Enamides".
J. Org. Chem. 2009, 74, 9528-9530. [doi]
24. Tang, W.*; Sarvestani, M.; Wei, X.; Patel, N.; Narayanan, B.; Nummy, L. J.; Byrne, D.; Lee, H.; Yee, N.; Senanayake, C. H. "Formation of 2-Trifluoromethylphenyl Grignard Reagent via Magnesium-Halogen exchange: Process Safety Evaluation and Concentration Effect".
Org. Proc. Res. Dev. 2009, 13, 1426-1430. [doi]
23. Han, Z.*; Song, J. J.; Yee, N. K.; Xu, Y.; Tang, W.; Reeves, J. T.; Tan, Z.; Wang, X.; Lu, B.; Krishnamurthy, D.; Senanayake, C. H. "Development of a Preparative-Scale Asymmetric Synthesis of (R)-p-Tolyl Methyl Sulfoxide for Use in a One-Pot Synthesis of a Drug Intermediate Containing a Trifluoromethyl-Substituted Alcohol Functionality".
Org. Proc. Res. Dev. 2007, 11, 605-608. [doi]
Publications during graduate and postdoctoral research:
22. Ito, E.; Frederick, M. O.; Koftis, T. V.; Tang, W.; Petrovic, G.; Ling, T.; Nicolaou, K. C.* "Structure toxicity relationships of synthetic azaspiracid-1 and analogs in mice".
Harmful Algae 2006, 5, 586-591.
21. Nicolaou, K. C.*; Koftis, T. V.; Vyskocil, S.; Petrovic, G.; Tang, W.; Frederick, M. O.; Chen, D. Y.; Li, Y.; Ling, T.; Yamada, Y. M. A. "Total synthesis and structural elucidation of azaspiracid-1. Final assignment and total synthesis of the correct structure of azaspiracid-1".
J. Am. Chem. Soc. 2006, 128, 2859-2872.
20. Nicolaou, K. C.*; Chen, D. Y.; Li, Y.; Uesaka, N.; Petriovic, G.; Koftis, T. V.; Bernal, F.; Frederick, M. O.; Govindasamy, M.; Ling, T.; Pihko, P. M.; Tang, W.; Vyskocil, S. "Total synthesis and structural elucidation of azaspiracid-1. Synthesis-based analysis of originally proposed structures and indication of their non-identiy to the natural product".
J. Am. Chem. Soc. 2006, 128, 2258-2267.
19. Nicolaou, K. C.*; Tang, W.; Dagneau P.; Faraoni, R. "A catalytic asymmetric three-component 1,4-addition/aldol reaction: Enantioselective synthesis of the spirocyclic system of vannusal A".
Angew. Chem., Int. Ed. 2005, 44, 3874-3879.
18. Nicolaou, K. C.*; Koftis, T. V.; Vyskocil, S.; Petriovic, G.; Ling, T.; Yamada, Y. M. A.; Tang, W.; Frederick, M. O. "Structural revision and total synthesis of Azaspiracid-1, part 2: Definition of the ABCD domain and total synthesis".
Angew. Chem., Int. Ed. 2004, 43, 4312-4318.
17. Nicolaou, K. C.*; Vyskocil, S.; Koftis, T. V.; Yamada, Y. M. A.; Ling, T.; Chen, D. Y.; Tang, W.; Petriovic, G.; Frederick, M. O.; Li, Y.; Sataki, M. "Structural revision and total synthesis of Azaspircad-1, part 1: Intelligence Gathering and tentative proposal".
Angew. Chem., Int. Ed. 2004, 43, 4318-4324.
16. Liu, D.; Tang, W.; Zhang, X.* "Synthesis of a new class of conformationally rigid phosphino-oxazolines: Highly enantioselective ligands for Ir-catalyzed asymmetric hydrogenation,".
Org. Lett. 2004, 6, 513-516.
15. Tang, W.; Wu, S.; Zhang, X.* "Enantioselective hydrogenation of tetrasubstituted olefines of cyclic β-(acylamino) acrylates".
J. Am. Chem. Soc. 2003, 125, 9570-9571.
14. Tang, W.; Wang, W.; Chi, Y.; Zhang, X.* "A P-chiral-bisphosphepine ligand for practical synthesis of β-aryl β-amino acids via asymmetric hydrogenation". Angew. Chem., Int. Ed. 2003, 42, 3509-3511.
13. Tang, W.; Zhang, X.* "New chiral phosphorous ligands for enantioselective hydrogenation".
Chem. Rev. 2003, 103, 3029-3069.
12. Tang, W.; Liu, D.; Zhang, X.* "Asymmetric hydrogenation of itaconic acid and enol acetate derivatives with the Rh-TangPhos catalyst".
Org. Lett. 2003, 5, 205-207.
11. Tang, W.; Wang, W.; Zhang, X.* "A new class of phospholane-oxazoline ligands for Ir-catalyzed asymmetric hydrogenation".
Angew. Chem., Int. Ed. 2003, 42, 943-946.
10. Ma, D.*; Xia, C.; Jiang, J.; Zhang, J.; Tang, W. "Aromatic nucleophilic substitution or CuI-catalyzed coupling route to Martinellic Acid".
J. Org. Chem. 2003, 68, 442-451.
9. Wu, S.; Wang, W.; Tang, W.; Lin, M.; Zhang, X.* "Highly enantioselective hydrogenation of enol acetates catalyzed by Ru-TunaPhos complexes".
Org. Lett. 2002, 4, 4495-4497.
8. Tang, W.; Zhang, X.* "Highly efficient synthesis of chiral beta-amino acid derivatives via asymmetric hydrogenation".
Org. Lett. 2002, 4, 4159-4161.
7. Korber, K.; Tang, W.; Hu, X.; Zhang, X.* "A practical synthesis of 2-amino-2'-hydroxy-1,1'-binaphthyl (NOBIN)".
Tetrahedron Lett. 2002, 43, 7163-7165.
6. Tang, W.; Chi, Y.; Zhang, X.* "An ortho-substituted BIPHEP ligand and its applications in Rh-catalyzed hydrogenation of cyclic enamides".
Org. Lett. 2002, 4, 1695-1698.
5. Tang, W.; Zhang, X.* "A chiral 1,2-bisphospholane ligand with a novel structural motif: Applications in highly enantioselective Rh-catalyzed hydrogenations". Angew. Chem., Int. Ed. 2002, 41, 1612-1614.
4. Zhou, Y.; Tang, W.; Wang, W.; Li, W.; Zhang, X.* "Highly effective chiral ortho-substituted BINAPO ligands (o-BINAPO): Applications in Ru-catalyzed asymmetric hydrogenations of beta-aryl-substituted beta-(acylamino)acrylates and beta-keto esters".
J. Am. Chem. Soc. 2002, 124, 4952-4953.
3. Tang, W.; Hu, X.; Zhang, X.* "A new chiral ruthenium complex for catalytic asymmetric cyclopropanation".
Tetrahedron Lett. 2002, 43, 3075-3078.
2. Ma, D.*; Tang, W.; Kozikowski, A. P.; Lewin, N. E.; Blumberg, P. M.; "General and stereospecific route to 9-substituted, 8,9-disubstituted, and 9,10-disubstituted analogues of benzolactam-V8".
J. Org. Chem. 1999, 64, 6366-6373.
1. Tang, W.; Ma, D.* "Stereospecific synthesis of 9-substituted benzolactam-V8 from L-tyrosine via orientation transfer of aromatic nitration".
Tetrahedron Lett. 1998, 39, 7369-7372.
|
Book Chapters:
4. Wan, F. Yang, H. Tang, W."Chiral Monophosphorus Ligand" of Full Book "Chiral Ligands: Evolution of Ligand Libraries for Asymmetric Catalysis". pa 233-260,2021.Linker
3. Li, K.; Tang, W."Air-stable P-chiral Phosphorus Ligands
for Asymmetric Catalysis and Synthesis" of Full Book "The Strem Chemiker". pa XXX-XXX,2019.Linker
2. Chi, Y.; Tang, W.; Zhang, X. "The other bisphosphine ligands for enantioselective alkene hydrogenation" in Book "Handbook of Homogeneous Hydrogenation". pa 853-882,Editor(s): Prof. Dr. Johannes G. de Vries and Prof. Dr. Cornelis J. Elsevier.
Wiley-VCH, Weinheim, Germany, 2007.
1. Chi, Y.; Tang, W.; Zhang, X. "Rhodium-catalyzed asymmetric hydrogenation". In Book "Modern Rhodium-Catalyzed Organic Reactions" pa 1-31,Editor: P. Andrew Evans, Wiley-VCH, Weinheim, Germany, 2005.
|
|
|
20. Tang, W.; Fu, W.; Liu, G. "Chiral diphosphine ligand, preparation method, intermediate and application thereof".
Patent No. CN 109293700 A Date Issued: February 1, 2019
19. Tang, W.; Si, T.; Li, B. "Benzo polyaza and phosphole oxygen ligand and complex containing same, preparation method and application".
Patent No. CN 109096338 A Date Issued: December 28, 2018
18. Tang, W.; Chen, D.; Xu, G. "Chirality 1,2-diamine compound and synthetic method thereof".
Patent No. CN 108929268 A Date Issued: December 4, 2018
17. Tang, W.; Jiang, W. "Biaryl diphosphine ligand as well as preparation method and application thereof".
Patent No. CN 107827929 A Date Issued: March 23, 2018
16. Tang, W.; Li, C.; Wan, F. "Metal complex as well as preparation method, application and intermediate thereof".
Patent No. CN 107445999 A Date Issued: December 8, 2017
15. Tang, W.; Zhao, G.; Hu, N. "Transition metal complexes and preparation of chiral α-amino boronic esters".
Patent No. CN 106146543 A Date Issued: November 23, 2016
14. Tang, W.; Huang, L.; Zhu, J. "Aryl substituted alcohols and synthetic methods of escitalopram".
Patent No. CN 105732249 A Date Issued: July 6, 2016
13. Tang, W.; Li, C. "Addressing the Challenging Issues in Crossing-Couplings by Ligand Design".
Patent No. CN 104650145 A Date Issued: May 27, 2015
12. Tang, W.; Liu, G. "Chiral phosphine ligand, metal catalyst comprising the chiral phosphine ligand and their application".
Patent No. CN 103087105 Date Issued: May 8, 2013
11. Schnaubelt, J.; Tang, W. "Method for stereoselective synthesis of 1,4-protected 9-hydroxy-5-oxo-1,4-diaza-spiro[5.5]undecanes".
Patent No. WO 2013117568 Date Issued: Aug 15, 2013
10. Haddad, N.; Qu, B.; Rodriguez, S.; Senanayake, C. H.; Tang, W.(as core maker); Wei, X.; Yee, N. K. "Monophosphorus ligands and their use in cross-coupling reactions".
Patent No. WO 2011126917 Date Issued: Oct 13, 2011
9. Qu, B.; Senanayake, C. H.; Tang, W.(as core maker); Wei, X.; Yee, N. K. "Novel chiral phosphorus ligands".
Patent No. WO 2011056737 Date Issued: May 12, 2011
8. Li, W.; De Croos, P.; Fandrick, K. R.; Gao, J. J.; Haddad, N.; Lu, Z.; Qu, B.; Rodriguez, S.; Senanayake, C. H.; Zhang, Y.; Tang, W. "A process for the preparation of quinoline derivatives for use as HIV integrase inhibitors".
Patent No. WO 2012138670 Date Issued: Oct 11, 2012
7. Patel, N. D.; Senanayake, C. H.; Tang, W.; Wei, X.; Yee, N. K. "Process for preparation of alkoxybromochloroarylquinolines from alkoxydichloroquinolines".
Patent No. WO 2010129451 Date Issued: Nov 11, 2010
6. Haddad, N.; Krishnamurthy, D.; Reeves, D. C.; Senanayake, C. H.; Tang, W.; Yee, N. K. "Process for preparation of (S)-3-[4-(benzyloxy)-3,5-dimethylphenyl]-2-[4-(2-oxo- 4,5-dihydro-1H-benzo[d][1,3]diazepin-3(2H)-yl)piperidine-1-carbonyloxy]propanoic aci and related compounds".
Patent No. WO 2010048138 Date Issued: Apr 29, 2010
5. Han, Z.; Kai, G.; Krishnamurthy, D.; Matthes, Burkhard.; Nar, H.; Priepke, H.; Schuler-Metz, A.; Senanayake, C. H.; Sieger, P.; Tang, W.; Wienen, W.; Xu, Y.; Yee, N. K. "Synthesis of 3-aminotetrahydrofuran-3-carboxylic acid derivatives for use as medicaments".
Patent No. WO 2008080891 Date Issued: Jul 10, 2008
4. Wang, X.; Sun, X.; Zhang, L.; Tang, W.; Krishanmurthy D.; Senanayake, C. H.; Han, Z. "Processes for the preparation of glucopyranosyl-substituted benzyl or benzene derivatives".
Patent No. WO 2006120208 Date Issued: Nov 16, 2006
3. Xumu, Z.; Tang, W. "Preparation of chiral cyclic amino acids and derivatives".
Patent No. US 2004242889 Date Issued: Dec 2, 2004
2. Xumu, Z.; Tang, W. "P-Chiral phospholanes and phosphocyclic compounds and their use in asymmetric catalytic reactions".
Patent No. US 2004229846 Date Issued: Nov 18, 2004
1. Xumu, Z.; Tang, W. "P-Chiral phospholanes and phosphocyclic compounds and their use in asymmetric catalytic reactions".
Patent No. WO 2003042135 Date Issued: May 22, 2003
|
@ 2015. The Tang Group. All rights reserved.
|